clinical
QUANTUM MOLECULAR RESONANCE
Dr Surbhi Virmani looks at the science behind this innovative regenerative technology
DR SURBHI VIRMANI
Dr Surbhi Virmani graduated in medicine in 1996 following which she was awarded a masters in anaesthesia, PG dip in clinical dermatology from London, PG dip in aesthetic medicine from London and bachelor of laws (LLB). She has 20 years of experience in anaesthetics and critical care working worldwide and for the NHS before moving exclusively to aesthetics and cosmetic dermatology. cosdermuk@gmail.com
Regenerative medicine, as we currently understand it, revolves around three core principles: cell-based therapies such as stem cell transplants, non-cell-based treatments like exosomes, and biophysical methods. The latter includes biochemical signals, biomechanical forces, and bioelectric communication.1 Significant progress has been made in understanding traditional cellular signalling, involving proteins, lipids, hormones, carbohydrates, RNA, and other small molecules that control cell fate decisions. However, the role of biophysical forces—mechanical, magnetic, acoustic, redox, and bioelectrical—is often underappreciated in shaping biology at both the cellular and tissue levels.2
Regeneration necessitates the precise coordination of growth and morphogenesis. This process involves a powerful yet largely enigmatic system of biophysical signals that operate during regeneration, embryonic development, and the formation of tumours. Ion transporters play a critical role by generating pH and voltage gradients, as well as ion fluxes, which in turn regulate cell proliferation, differentiation, and migration. Endogenous bioelectrical signals are crucial for controlling wound healing, limb development, left-right patterning, and spinal cord regeneration. Recent breakthroughs in molecular biology and imaging technology have provided unprecedented insights into the sources and downstream effects of ion flows. Beyond the current emphasis on molecular genetics and stem cell biology, the artificial modulation of bioelectrical signals in somatic tissues represents a powerful approach that could significantly enhance our understanding and augmentation of regenerative capacity.
From a quantum biology perspective, electromagnetic interactions and photonic communications at the intracellular and intercellular levels are essential for the evolving complexity of eukaryotic cells and metazoan species.
DOES ELECTRICITY PLAY A ROLE IN OUR CELLS?
All cells possess ion-channel proteins and pumps that establish their resting potential (Vmem), a key parameter regulating various cell behaviours, including proliferation, differentiation, apoptosis, migration, and polarisation. This phenomenon is observed across a wide range of cells, from yeast to human stem cells, and is vital for processes such as stem cell differentiation and embryo development.3
The transmembrane potentials of eukaryotic cells, regulated by ion channels and pumps, are crucial for embryo development and organ morphogenesis. Ion channels allow passive ion flow across the cell membrane along electrical and concentration gradients, while ion pumps work against these gradients, often coupled with ATPase activities.4,5
Research on model organisms, including planarians, drosophila, zebrafish, xenopus, mice, and humans with specific genetic mutations in ion channels and pumps, indicates that changes in transmembrane potentials can lead to significant morphological alterations during development. 6,7, 8,9, 10,11,12
There is growing scientific interest in the roles of specific ion channels in development, cancer, and regeneration, and in the mechanisms of endogenous bioelectricity in wound healing and regeneration.13,14,15
An organelle of interest here is the centrosome, which is an organelle in eukaryotic cells, and functions as a nanoscale molecular electronic generator that continuously supplies the microtubule network with electric currents, creating an electromagnetic field that facilitates mitosis.16
Electromagnetic oscillations in chromatin, triggered by the energy-consuming activities of DNA/RNA polymerases and cytoskeletal electronic pulses, are transmitted to chromatin fibres through microtubules, generated by the centrosome and the cytoskeleton ATPases. These interactions regulate gene transcription and tissue morphogenesis.17, 18, 19
During embryogenesis, cell proliferation and differentiation result in programmed changes in cellular electromagnetic fields (oscillatory frequencies and bioelectricity of transmembrane potentials) across different parts of the embryo, leading to organ formation.11
The electromagnetic field of cells in a specific organ directs stem cells and progenitor cells when to differentiate or self-renew by influencing their chromatin organisation.20,21,22
The preference for specific cellular electromagnetic frequencies to differentiate or remain quiescent determines the size and shape of tissues. Epigenetic regulatory loops, triggered by changes in the electromagnetic oscillation frequencies of chromatin regions rich in noncoding RNA, play a role in these processes.
Michael Levin proposed that bioelectricity, regulated by transmembrane potentials, can be seen as the software that programmes the cellular hardware to perform complex morphogenetic transformations.
SELECTIVE STIMULATION OF CELLS CAPABLE OF REGENERATION
Membrane potentials of cells from more differentiated states are usually hyperpolarised.23 Thus, they are much less sensitive to the environmental changes of EMF, and the intracellular EMF of differentiated cells is not likely to be interfered with by the alterations of EMF of other cells.24 Alternatively stem cells and progenitor cells usually possess depolarised membrane potential, which is more likely to transcriptionally respond to the changes of external EMF through the alteration of intracellular EMF.11,24
Crucially, it is becoming clear that bioelectricity is not simply one more layer of mechanism that is required alongside biochemical cues and stress forces to implement morphogenesis: it enables unique, powerful information-processing capacity that facilitates scaling of cells into complex morphogenetic collectives.
It is important to remember that, in humans, bioelectric signals can regulate the electro diffusion of neurotransmitter molecules especially serotonin in and out of cells, alter clustering of KRAS receptors in the membrane,25 influence voltage-sensitive phosphatases,26 modulate integrin signaling,27 regulate cell volume,28 and alter chromatin states.29 The results of the bioelectric currents delivered during the developmental phases can be seen long-term or after a delayed period in the outcome.
In regeneration, the bioelectric circuits tend to function as the early decision-making switch that selects among downstream morphogenetic programmes that function long after the initial stimulus is gone. For example, prolonged osteogenic effects occur long after discontinuing electrical stimulation,30 and the post eight to 10 days of regeneration of planarian heads and tadpole tails is kickstarted by an electrical state occurring during three hours or one hour, respectively during their development. 31, 32
UNDERSTANDING QMR
In physics, a quantum refers to a discrete amount of energy that is directly proportional to the frequency of the associated radiation. For example, a photon represents a quantum of light, and an electron represents a quantum of electricity. According to Planck’s law, the energy delivered depends solely on the frequency.
The term “quantum” was introduced to modern physics by Max Planck in 1901. While attempting to explain black-body radiation and the colour changes of heated objects, Planck proposed that energy is not emitted in a continuous wave but in discrete packets or bundles, which he called quanta.
The relationship between energy and frequency is captured in the equation E=hf, also called the Planck relation or PlanckEinstein relation, where E is energy, h is Planck’s constant, and f is the frequency of the electromagnetic wave. Essentially, this means that the energy generated is proportional to the frequency of the wave.
AQMR wave comprises specially tuned peaks of multiple electromagnetic frequencies, specifically 16 peaks at 4MHz intervals ranging from 4 to 64MHz. This configuration stimulates natural cellular metabolism by delivering energy directly to skin cells and mesenchymal stem cells (MsC).
Beautyform Medical’s Rexonage3 technology is built upon the scientific principles detailed above, delivering innovative regenerative treatments through Quantum Molecular Resonance (QMR). Rexonage3 utilises this precise frequency modulation to enhance cellular activities, promoting regeneration and healing through the principles of quantum molecular resonance.
QMR devices generate the necessary quanta by altering harmonics, targeting biological tissue with precision. Each quantum acts like a bullet, breaking different molecular bonds through hyperresonance generated by the energy. This process disrupts molecular bonds without generating heat or causing trauma, resulting in the creation of free ions.
The creation of free ions leads to the modification of ionic concentration in the intracellular/extracellular matrix of the cells, generating a significant change in the transmembrane potential of the target cells.
WHAT IS THE OUTCOME OF THE CHANGE OF THIS TRANSMEMBRANE POTENTIAL?
1. The primary action of this device is to target the mesenchymal stem cells (MSCs) located under the skin, often found in fat pads that diminish as we age. MSCs are multipotent adult stem cells capable of differentiating into various cell types within the mesodermal lineage.
Using QMR technology, cell regeneration is stimulated by enhancing cell metabolism. This stimulation leads to fat regeneration, skin regeneration, and remodeling of the basal membrane and dermis.
QMR applied to human mesenchymal stromal cells from bone marrow showed transcriptional changes and up regulation of genes (e.g., MMP1, PLAT, A2M) which are involved in the development processes of embryogenesis, wound healing and angiogenesis.33
2. The second effect of QMR technology is its ability to decrease inflammation and mitigate the inflammatory component of ageing. When QMR is applied to tissue, it polarises resident macrophages, which then produce anti-inflammatory cytokines such as IL-10 and reduce the levels of inflammatory cytokines like IL-1 . This unique capability makes QMR technology particularly effective in addressing the inflammatory aspects of the ageing process.
3. QMR technology significantly enhances the production of collagen type III, which is crucial for both structural support and cellular function. Collagen type I is a stiff fibrillar protein that provides tensile strength, whereas collagen type III forms an elastic network, storing kinetic energy through elastic rebound.
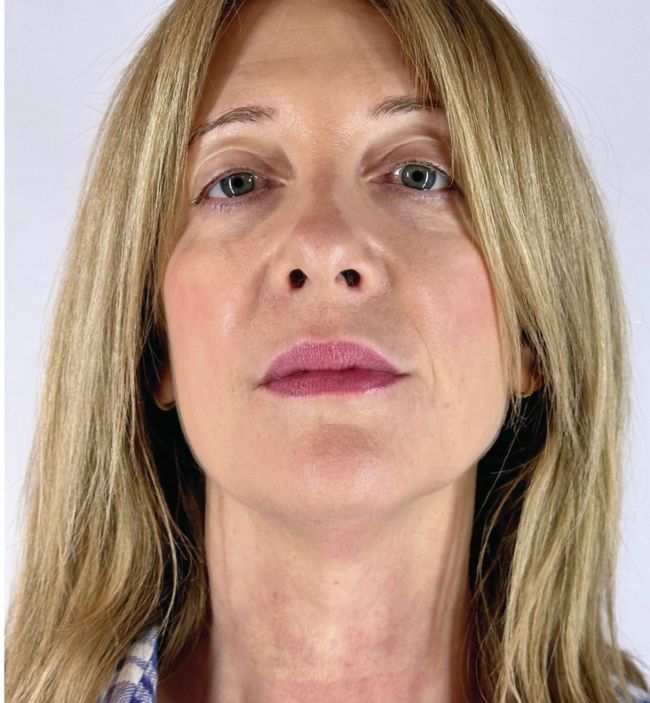
Dr SV Rexonage3 November 2023 April 2024
Collagen type I builds a scaffold with thick fibres, but its maturation depends on collagen type III, which generates thinner, less durable fibres with high turnover. This interplay between collagen types I and III maintains the structural integrity and elasticity of tissues, emphasising collagen type III’s role in regeneration. Collagen type I is found in skin, tendons, vasculature, lungs, heart, and other organs, and it constitutes most of the organic material in bones and teeth. Reticular fibres, composed of collagen type III, often accompany collagen type I.
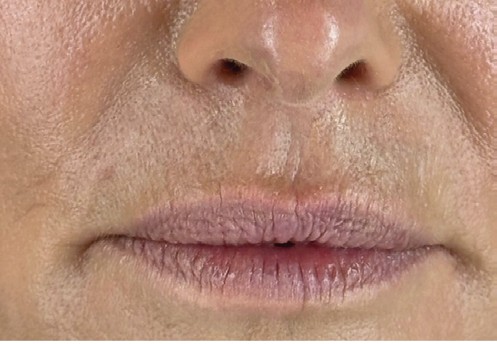
Dr SV Rexonage lips
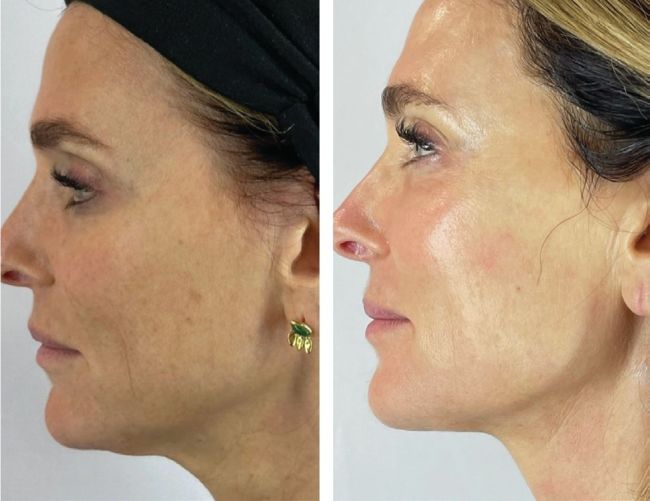
Dr SV rexonage and combination treatments with profhilo, PRP and obagi nuderm system
LIMITATIONS OF HEAT-BASED TECHNOLOGIES
At temperatures significantly above 37°C, collagen’s triple-helix structure collapses as both interand intramolecular bonds are broken, leading to the dissociation of the resulting unstructured collagen molecules. Due to time-temperature equivalence, denaturation does not occur at a specific temperature but rather at specific time-temperature combinations. When a critical time-temperature threshold is exceeded, the functional collagen is eventually lost, transforming the tissues into gel-like, gelatinous materials. This transformation results in a loss of load-bearing ability, including stiffness and toughness. Additionally, soft tissues shrink significantly during denaturation. These processes are not instantaneous but instead follow exponential functions.36
LONG-LASTING EFFECTS
One of the most significant advantages of the QMR treatment is its long-lasting effects. Studies have shown that the application of bioelectrical signals not only provides immediate results but also leads to delayed outcomes. Consequently, patients will continue to experience improvements in the months following the treatment. The treatment is not based on heat or inflammation, it is not limited by colour of the skin and there is no downtime or pain for the patient. It is regenerative at all levels by stimulating mesenchymal stem cells and reducing the inflammatory component of ageing, and the likelihood of dealing with patient complaints related to scarring, post-inflammatory hyperpigmentation, fat loss and many more (rare but not impossible) does exist.35,36

REFERENCES
1. Levin M. Bioelectric signalling: Reprogrammable circuits underlying embryogenesis, regeneration, and cancer.
2. Sun G, Li J, Zhou W, Hoyle RG, Zhao Y. Electromagnetic interactions in regulations of cell behaviors and morphogenesis.
3. BlackistonD, McLaughlin K, Levin M. Bioelectric controls of cell proliferation: ion channels, membrane voltage and the cell cycle.
4. Takeuchi A, Reyes N, Artigas P, Gadsby DC. Visualizing the mapped ion pathway through the Na,K-ATPase pump.
5. Gadsby DC, Takeuchi A, Artigas P, Reyes N. Review. Peering into an ATPase ion pump with single-channel recordings.
6. Adams DS, Uzel SG, Akagi J, Wlodkowic D, Andreeva V, Yelick PC, Devitt-Lee A, Pare JF, Levin M. Bioelectric signalling via potassium channels: a mechanism for craniofacial dysmorphogenesis in KCNJ2-associated Andersen-Tawil Syndrome.
7. George LF, Pradhan SJ, Mitchell D, Josey M, Casey J, Belus MT, Fedder KN, Dahal GR, Bates EA. Ion Channel Contributions to Wing Development in Drosophila melanogaster.
8. Pai VP, Pietak A, Willocq V, Ye B, Shi NQ, Levin M. HCN2 Rescues brain defects by enforcing endogenous voltage pre-patterns. 9. George LF, Pradhan SJ, Mitchell D, Josey M, Casey J, Belus MT, Fedder KN, Dahal GR, Bates EA. Ion Channel Contributions to Wing Development in Drosophila melanogaster.
10. Lanni JS, Peal D, Ekstrom L, Chen H, Stanclift C, Bowen ME, Mercado A, Gamba G, Kahle KT, Harris MP. Integrated K+ channel and K+Clcotransporter functions are required for the coordination of size and proportion during development.
11. Levin M. Bioelectric signaling: Reprogrammable circuits underlying embryogenesis, regeneration, and cancer.
12. Pezzulo G, LaPalme J, Durant F, Levin M. Bistability of somatic pattern memories: stochastic outcomes in bioelectric circuits underlying regeneration.
13. Bates E. Ion channels in development and cancer.
14. McLaughlin KA, Levin M. Bioelectric signaling in regeneration: Mechanisms of ionic controls of growth and form.
15. Silver BB, Nelson CM. The Bioelectric Code: Reprogramming Cancer and Aging From the Interface of Mechanical and Chemical Microenvironments.
16. Nygren J, Adelman RA, Myakishev-Rempel M, Sun G, Li J, Zhao Y. Centrosome as a micro-electronic generator in live cell.
17. Zhao Y, Zhan Q. Electric oscillation and coupling of chromatin regulate chromosome packaging and transcription in eukaryotic cells.
18. Pliss A, Malyavantham KS, Bhattacharya S, Berezney R. Chromatin dynamics in living cells: identification of oscillatory motion.
19. Niekamp S, Coudray N, Zhang N, Vale RD, Bhabha G. Coupling of ATPase activity, microtubule binding, and mechanics in the dynein motor domain.
20. Ross CL, Siriwardane M, Almeida-Porada G, Porada CD, Brink P, Christ GJ, Harrison BS. The effect of lowfrequency electromagnetic field on human bone marrow stem/progenitor cell differentiation.
21. Maziarz A, Kocan B, Bester M, Budzik S, Cholewa M, Ochiya T, Banas A. How electromagnetic fields can influence adult stem cells: positive and negative impacts.
22. Suryani L., Foo J. K. R., Cardilla A., Dong Y., Muthukumaran P., Hassanbhai A., (2021). Effects of pulsed electromagnetic field intensity on mesenchymal stem cells.
23. Numaga-Tomita T., Shimauchi T., Oda S., Tanaka T., Nishiyama K., Nishimura A., (2019). TRPC6 regulates phenotypic switching of vascular smooth muscle cells through plasma membrane potential-dependent coupling with PTEN.
24. Li X., Yang F., Rubinsky B. (2021). A correlation between electric fields that target the cell membrane potential and dividing HeLa cancer cell growth inhibition.
25. Zhou Y, Wong CO, Cho KJ, van der Hoeven D, Liang H, Thakur DP, Luo J, Babic M, Zinsmaier KE, Zhu MX, Hu H, Venkatachalam K, Hancock JF. SIGNAL TRANSDUCTION. Membrane potential modulates plasma membrane phospholipid dynamics and K-Ras signaling.
26. Numaga-Tomita T., Shimauchi T., Oda S., Tanaka T., Nishiyama K., Nishimura A., (2019). TRPC6 regulates phenotypic switching of vascular smooth muscle cells through plasma membrane potential-dependent coupling with PTEN.
27. Becchetti A, Crescioli S, Zanieri F, Petroni G, Mercatelli R, Coppola S, Gasparoli L, D’Amico M, Pillozzi S, Crociani O, Stefanini M, Fiore A, Carraresi L, Morello V, Manoli S, Brizzi MF, Ricci D, Rinaldi M, Masi A, Schmidt T, Quercioli F, Defilippi P, Arcangeli A. The conformational state of hERG1 channels determines integrin association, downstream signaling, and cancer progression.
28. Yellin F, Li Y, Sreenivasan VKA, Farrell B, Johny MB, Yue D, Sun SX. Electromechanics and Volume Dynamics in Nonexcitable Tissue Cells.
29. Matzke AJM, Lin WD, Matzke M. Evidence That Ion-Based Signaling Initiating at the Cell Surface Can Potentially Influence Chromatin Dynamics and ChromatinBound Proteins in the Nucleus.
30. Eischen-Loges M, Oliveira KMC, Bhavsar MB, Barker JH, Leppik L. Pretreating mesenchymal stem cells with electrical stimulation causes sustained long-lasting pro-osteogenic effects.
31. Durant F, Bischof J, Fields C, Morokuma J, LaPalme J, Hoi A, Levin M. The Role of Early Bioelectric Signals in the Regeneration of Planarian Anterior/Posterior Polarity.
32. Tseng AS, Beane WS, Lemire JM, Masi A, Levin M. Induction of vertebrate regeneration by a transient sodium current.
33. Sella S, Adami V, Amati E, Bernardi M, Chieregato K, Gatto P, Menarin M, Pozzato A, Pozzato G, Astori G. In-vitro analysis of Quantum Molecular Resonance effects on human mesenchymal stromal cells.
34. Fraccalvieri M, Salomone M, Di Santo C, Ruka E, Morozzo U, Bruschi S. Quantum molecular resonance technology in hard-to-heal extremity wounds: histological and clinical results.
35. Hitchcock TM, Dobke MK. Review of the safety profile for microfocused ultrasound with visualization. 36. Meador WD, Sugerman GP, Tepole AB, Rausch MK. Biaxial mechanics of thermally denaturing skinPart 1: Experiments.