Commercial Feature
Refresh and revive
Dr Alexis Granite discusses treatment of a 52–year–old patient combining BELOTERO ® Revive and Volume
Dr Alexis Granite is a consultant dermatologist at Skinesis Medical at Sarah Chapman in London. She is also American Board certified in Dermatology. With over 14 years of clinical experience, Dr Granite specialises in both aesthetics and medical dermatology. Dr Granite is a member of the British Association of Dermatologists (BAD), the American Academy of Dermatology (AAD) and the British Cosmetic Dermatology Group (BCDG). She is quoted extensively in the press and has published numerous articles in several peer-reviewed journals, as well as authored book chapters on dermatologyrelated topics.
INTRODUCTION
Our understanding of the ageing process has advanced significantly over the last decade. Ageing involves profound changes not only in the skin and subcutaneous tissue but also in facial bone, muscle and ligaments.
Addressing this multitude of changes is vital to providing aesthetic patients with the most natural and harmonious treatment results. Increasingly, patients desire subtle, refreshed improvements in their appearance without looking “done”. Because no single treatment modality can target all of the underlying processes in facial ageing, combining modalities provides optimal outcomes.
In my approach to aesthetic patients as a dermatologist, I feel skin health involves a 360°, holistic approach encompassing overall wellness, a robust skincare routine, management of any underlying skin conditions and in-clinic treatments.
Now, more than ever, patients seek to wear minimal make-up, instead preferring to focus on skin quality as an indicator of youthfulness and wellbeing.
BELOTERO ® REVIVE AND COMBINATION TREATMENTS
I have found skin revitalisers such as BELOTERO ® Revive, designed to enhance the quality of photodamaged skin1, to be an incredibly versatile treatment with a high patient satisfaction rate.1 BELOTERO Revive is easily combined with other aesthetic modalities such as dermal fillers, plateletrich plasma and machine-based therapies such as laser, high-intensity focused ultrasound (HIFU) and radiofrequency (RF) as part of a comprehensive rejuvenation plan over multiple visits.
BELOTERO ® Revive can be combined with other injectables in the same treatment session when used over different facial areas and is also an excellent option for younger patients with early signs of photodamage1 as an introduction to aesthetic treatments.
BELOTERO ® Revive is comprised of 20mg/ ml cross-linked hyaluronic acid and 17.5mg/ mL glycerol.1 Hyaluronic acid (HA) boosts skin hydration1 and increases collagen and elastin production via fibroblast stimulation.3 HA has been demonstrated to rejuvenate skin across multiple clinical studies. And as with all of the products in the BELOTERO range, the use of Cohesive Polydensified Matrix (CPM ) technology allows for optimal, homogeneous tissue integration within the dermal matrix.3
The addition of glycerol and trihydroxy alcohol is a leading feature of BELOTERO ® Revive compared to other skin revitalisers. Glycerol enhances the potency of hyaluronic acid by strengthening the hyaluronic acid matrix 4 and boosting skin hydration and elasticity. 2
PATIENT ASSESSMENT
In the following case, this fit and well 52-yearold patient presented with a desire to improve her sagging jawline and freshen her skin’s overall appearance. On physical examination, she had slight hollowing over her temples, skin laxity along her jawline with early jowling, along with some fine lines and pore dilation, more prominent over the mid-face.
PRODUCT SELECTION
For this patient, I chose BELOTERO ® Volume5 with lidocaine for her temples and restoration of facial volume along the jawline, and BELOTERO Revive for overall facial skin hydration and revitalisation.
COMBINATION TREATMENT TECHNIQUE
Treatment was carried out with both needle and cannula injection techniques in a single sitting. First, a supraperiostal bolus of 0.25cc of BELOTERO ® Volume with lidocaine was placed within each temple using a 30G needle after aspirating for 10 seconds. The injection site was located 1cm inferior to the temporal infusion line and one finger’s breadth above the superior border of the zygoma.
BELOTERO ® Volume was then deposited as a bolus at the angle of the mandible and as linear threads along and just superior to each mandible using a 0.5 x 50mm 25G cannula via a single entry point for a total of 0.5cc on each side. Overall, 1.5ccs of BELOTERO ® Volume was used for this patient.
I then deposited small boluses of 0.05- 0.1cc of BELOTERO ® Revive within the mid-superficial dermis of the lateral forehead (two injection sites), periorbital region (one injection site) and lateral chin (one injection site) using a 30G needle. Lastly, an entry point was made in the mid-cheek, and BELOTERO Revive was then fanned throughout all four quadrants of the cheek within the midsuperficial dermis using a 0.5 x 50mm 25G cannula via the single entry point. A total of 2ccs was used.
The patient was consented and cleansed with chlorhexidine solution prior to the procedure. No topical anaesthetic was applied but given the lidocaine within BELOTERO ® Volume5 and the small needle gauge utilised, the patient was comfortable throughout. Transient erythema was noted immediately following injection, which settled within several hours. Bruising is a potential side effect of any injectable treatment, and the patient was given Arnica gel to apply twice daily at home as needed.
RESULTS
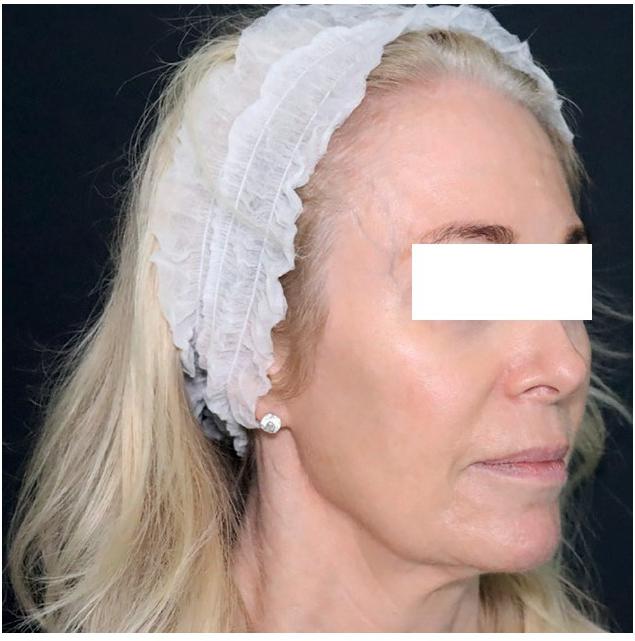
Before
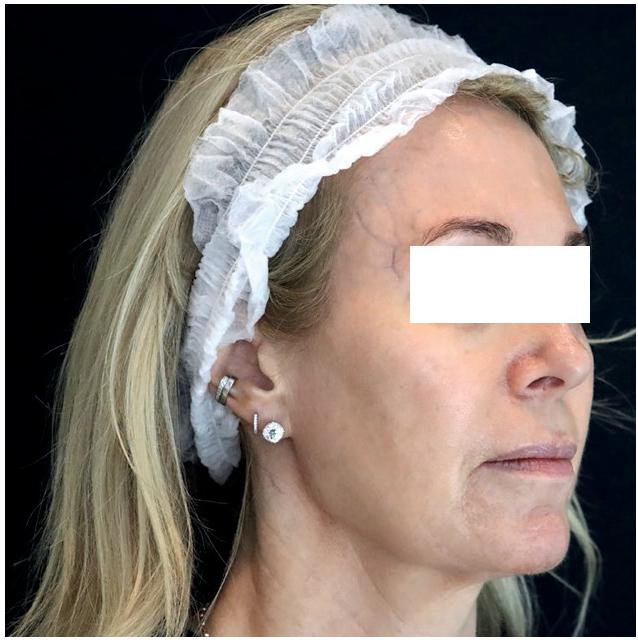
After
The patient was seen two weeks following her procedure, at which time an improvement in skin hydration, texture, pore size and skin laxity, in particular along the jawline, was noted. She was very pleased with her results and the improvement in skin quality.
BELOTERO ® Volume treatment results can be expected to last 12-18 months.6 The Merz Aesthetics BELOVE study demonstrated that improvements in skin hydration and pigmentation from BELOTERO Revive last up to nine months, improvements in skin softness up to seven months, and skin firmness improvements six months.2Additional sessions with BELOTERO Revive may be performed four and eight weeks following the initial treatment if needed.
Adverse events should be reported. Reporting forms and information for United Kingdom can be found at www.mhra.gov.uk/ yellowcard. Reporting forms and information for Republic of Ireland can be found at https://www.hpra.ie/homepage/about-us/ report-an-issue/mdiur. Adverse events should also be reported to Merz Pharma UK Ltd by email to UKdrugsafety@merz.com or on +44 (0) 333 200 4143.
This advertorial is sponsored by Merz Aesthetics UK & Ireland M-BEL-UKI-1212 Date of Preparation July 2021
REFERENCES
1. BELOTERO Revive IFU 2018
2. Belove. Clinical, Cosmetic and Investigational Dermatology 2019:12 563–572
3. Wang 2007. Arch Dermatol. 2007;143:155-163
4. Succi (2011) Rejuvenation of Periorbital Area. Dermatol Surg 2012;38:192–198
5. BELOTERO Volume IFU 2020
6. Micheels P, Vandeputte J, Kravtsov M. Treatment of Age-related Mid-face Atrophy. J Clin Aesthet Dermatol. 2015;8(3):28–34
For more information visit merz–aesthetics.co.uk, @merzaesthetics.uki (Instagram) or Merz Aesthetics UK & Ireland (Facebook)