Industry news
NEWS AND ANALYSIS
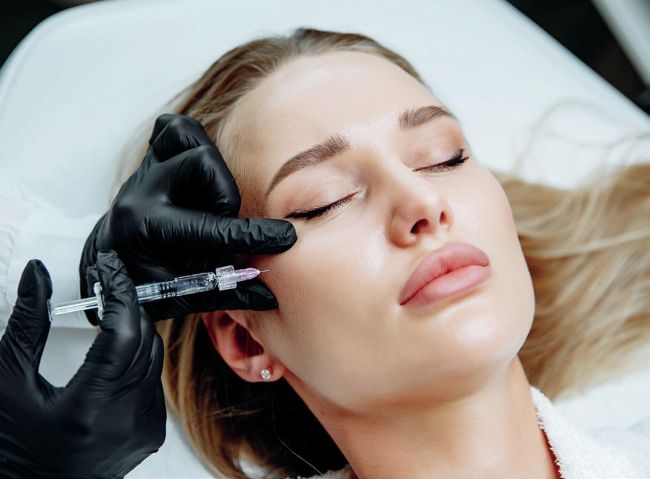
TikTok becomes research tool for cosmetic procedures, with lip filler most viewed hashtag
Users of TikTok are turning to the video-sharing app to research non-surgical procedures in huge numbers.
The hashtag “lip filler” is most popular, with over 1.8 billion views worldwide as of 23 July 2021, found new research from ExpressDentist.
The findings revealed the top ten aesthetic procedure and beauty treatment-related hashtags viewed on TikTok:
1. Lip filler – 1,872,500,000 views
2. Microblading – 918,600,000 views
3. Brow lamination – 446,000,000 views
4. Lip blushing – 118,800,000 views
5. Thread lift – 118,600,000 views
6. Non-surgical butt enhancements – 64,700,000
7. Non-surgical rhinoplasty – 55,600,000
8. Teeth whitening – 46,100,000
9. Non-surgical face lift – 25,500,000
10. Jawline contouring – 15,100,000
TikTok creators use tags like “#lipfiller” and “#lipfillercheck” to help other users find their videos, with one sound created to show off lip transformations before and after filler having over 253,000 videos. The app released a Big Lip filter for videos to imitate an overenhanced and animated suggestion of what it would look like if users had filler.
Thread lifts also ranked highly after the “fox eye” look went viral on the app. Using threads to lift the upper eyelid and brow tail became a trend as users discovered the long-term alternative to using make-up and shaving their eyebrows to achieve a look popularised by models Kendall Jenner and Bella Hadid and actress Megan Fox. Semi-permanent make-up treatments such as lip blushing and microblading were also highly searched for.
Anticipated Canadian dermal filler Revanesse arrives in the UK with nurse advisory board
A new HA dermal filler product is to officially launch onto the UK market in October. FDA-approved Revanesse, from Canadian medical technology company Prollenium, uses smooth, biocompatible spherical particles with low levels of modification, which the manufacturer says reduce irritation, pain and swelling due to their smaller surface area, while holding “the most amount of volume”.
The filler also undergoes a purification process to remove floating BDDE and impurities after being wet milled and sieved to produce a uniformly cross-linked gel that is designed to degrade at an even rate. Available in 1.2ml syringes, the range includes four variants with different sphere sizes for multiple indications: Kiss, for the lips; Contour, for the deep dermis to subcutaneous layers; Shape, for the supra-periosteum; and Ultra, for the mid to deep dermis. Revanesse is supported by a nurse advisory group led by Sharon Bennett, aesthetic nurse practitioner and chair of the British Association of Cosmetic Nurses (BACN). She said: “Safety and performance are key for me. The fact that Revanesse has received FDA approval in the US gives me reassurance that it is a safe and reliable product.”
Improved version of Swiss–made robotic syringe for toxin injections comes to market
Swiss medical technology company Juvaplus has created a robotic syringe to provide a safer alternative to manual injection of botulinum toxin.
CE-marked Juvapen Expert has been designed to streamline and perfect botulinum toxin injections, claiming to offer clinicians accuracy and precision that has previously been nearly impossible to achieve. The light and flexible robotic syringe, which is customisable for each application, reduces the margin of error when injecting small doses of toxin, known as microdosing or “baby botox”.
Juvapen Expert’s internal mechanisms produce a constant homogenous flow so that the toxin distributes evenly across the designated area, reducing pain, discomfort and visible swelling.
Aesthetic plastic surgeon and Aesthetic Medicine editorial board member Mr Ali Ghanem uses the device. He said: “It has become an integral part of my daily practice. My patients are both impressed and amazed by the difference in comfort this high-tech piece of equipment makes to their injectable experience, especially when I explain the added benefits with regard to its precision in dosing and anatomy.
“All of my patients claim the ‘robot’ – as we nickname it – has a lighter touch and, in many cases, have stated that it is completely painless compared to manual injections. For these reasons I use it routinely now. Overall, I believe it’s a far more patient-friendly way to administer botulinum toxin and other injectables for the most accurate results.”
Juvapen first launched in 2016 with the Juvapen Classic model. It has since been altered and the technology updated for re-release as Juvapen Expert.
Dija Ayodele to release book on Black skin
Aesthetic therapist, founder of West Room Aesthetics and Aesthetic Medicine editorial board member Dija Ayodele is to publish a book.
Black Skin: The Definitive Skincare Guide will be published by HarperCollins as a fully illustrated “inspirational skincare guide” for Black women, along with contextual social and historical insights into beauty and what it means to be a Black woman.
In the book, Ayodele will share education and advice around things like skincare ingredients and common skin complaints affecting darker skin tones. She will also seek to dispel myths and misconceptions surrounding Black skin in the beauty industry. Commenting on the announcement of the book, she said: “It’s been an absolute honour and joy to write Black Skin and I’m thrilled to finally be sharing my knowledge and experience with the world.
“My mission has always been to ensure all Black women are seen in skincare and over the last ten years, I’ve witnessed the anxiety that Black women experience in accessing information and treatments to care for their unique skincare needs. It stops here! Like me, this guide is sassy, fun, and plain speaking. I am really proud of Black Skin and excited to be putting skincare for Black women on the map in a way that’s never been done before.”
Black Skin: The Definitive Skincare Guide will be released on 25 November 2021.

NEWS IN BRIEF
Wigmore announces further training dates
Wigmore Medical has released its final PRP training course dates for 2021 in partnership with Croma-Pharma – 14 September, 21 October, 16 November and 14 December. The course covers the scientific principles, anatomy, clinical studies and best practice of PRP on the face and scalp to achieve optimum results for patients. It is suitable for doctors, nurses and dentists. Places on the course can be booked for £500. To book: wigmoremedical.com
SkinTech launches specialist peels training
SkinTech Pharma together with Med & Skin will hold three peels courses in September and October. The 13 September date will focus on skin anatomy physiology and superficial peels, while on 5 October the course will cover advanced training on TCA medium peels and combining TCA with phenol; and the final course will cover performing deep local peels with various phenol peels, on 11 October. Contact: info@med-skin.uk
Croma–Pharma secures Pliaglis licensing agreement
A nine-country licensing agreement has been made between Croma-Pharma and Crescita Therapeutics for topical local anaesthetic cream Pliaglis. It will be promoted directly to doctors through Croma’s sales network across the UK, Germany, Brazil and six more territories. “We are looking forward to collaborating with Crescita, an innovation-driven dermatology company on Pliaglis, which perfectly complements our portfolio of aesthetic injectable products, said Croma managing director Andreas Prinz.
Ivanmed to hold Dermapen 4 launch event in London
Aesthetics equipment supplier Ivanmed is to hold a launch event for the new Dermapen 4 at Doubletree by Hilton Tower of London on 11 and 12 September. The device is a digital one-touch integrated workstation for advanced microneedling with anticontamination management and a scar treatment setting, among other functions. Global trainer Dr Andrew Christie will be in attendance to share his knowledge and expertise. To attend: info@ivanmed.com
Campaign launches to allow images of nipple tattooing on social media sites
Medical micropigmentation artist and head medical micropigmentation trainer at Finishing Touches Group, Kelly Forshaw Smith, is to campaign Facebook over its social media sites Facebook and Instagram banning accounts sharing of images of nipple reconstructions.
Forshaw Smith specialises in nipple and areola reconstruction through tattooing, and has treated over 8,000 women and men, many of them following breast surgery due to cancer. She says herself and many of her peers have been banned from Facebook and Instagram countless times for posting images of their work because the pictures go against the sites’ community guidelines under the category of “sexual content and nudity”.
On 1 September Forshaw Smith and fellow micropigmentation artists will take to Facebook HQ in London dressed in inflatable breast costumes in attempt to persuade the company to make a change to its rules that would help empower breast cancer survivors to know that nipple and areola restoration treatments are available.
She commented: “...I have been banned from Facebook and Instagram countless times over the years, ranging from just having the image removed to not being able to post for over a week.”
Doctors and healthcare staff contributing to cybersecurity risks
Doctors may unknowingly be contributing to major online security risks thanks to “poor cybersecurity hygiene”, according to cybersecurity company NordVPN. It said that in 2020, ransomware attacks –a type of malicious software that threatens the user with publishing sensitive data or blocking access to a computer system unless a fee is paid – affected an estimated 600 separate healthcare institutions and more than 18 million individual patient records in 2020. This was a 470% increase from 2019.
The majority of these security incidents are a result of employee negligence and simple human error, said the company, such as weak password management, unsecure data storing and sharing and using out-ofdate software. Doctors and healthcare staff in GP surgeries and other clinical settings are being advised to implement easy-tofollow measures. These include creating complex and unique passwords for all online accounts and storing them safely in a password manager, encrypting patient data and medical files, using a VPN (virtual private network) for a safe internet connection; and educating themselves on cybersecurity, such as how to identify phishing emails.
Study shows effectiveness of Nd:YAG on basal cell carcinoma
A new clinical study has found that a combination of Nd:YAG laser and optical coherence tomography (OCT) imaging can provide an effective non-invasive treatment for the majority of basal cell skin cancers.
In the clinical report, Combining Nd:YAG laser with optical coherence tomography for nonsurgical treatment of basal cell carcinoma, 102 patients with 119 basal cell caricnomas were treated. The 1064nm Nd:YAG laser was applied and tumour margins measured using OCT. Patients received treatments every two months until no residual tumors remained and were monitored for one year afterwards. Results showed that 71.4% of tumours were cleared after just one session, while the rate of complete clearance without visible recurrence in all patients was 98%.
Aesthetic industry names weigh in on APPG recommendations
Following the report released in July by The All Party Parliamentary Group on Beauty, Aesthetics and Wellbeing (APPG BAW) which called on the UK Government to address the absence of regulation over botulinum toxin, dermal fillers and other non-surgical procedures, many leading industry names have commented on the recommendations.
The Group’s report came after a year-long inquiry into the issue due to the explosion in the popularity and availability of aesthetic treatments, as well as concern around the lack of a legal framework of standards, which the APPG says – echoing years of outrage and frustration from the medical aesthetic community – leaves consumers at risk and undermines the field’s ability to develop.
The Group, chaired by Carolyn Harris MP and Judith Cummins MP, made 17 recommendations for Government to fill the regulatory gap, including introducing a national licensing framework and making fillers prescription-only. These recommendations are based on evidence given in public inquiry sessions and written submissions from a range of stakeholders.
Commenting on the report, Dr Tristan Mehta, founder of clinic STORY and Harley Academy – which devised a Level 7 Diploma in Botox and Dermal Fillers in partnership with the JCCP to give healthcare professionals access to best-in-class medical aesthetics training – said: “…Through calling for fillers to become prescription-only and mandatory medical oversight for such treatments, the risk of permanent complications can be significantly reduced – the treatment for many complications from injectables needs a medical prescriber.” He added:
“Some skeptics may see this as just another report and recommendation, but when will this become legislation? It is likely that regulation will be enforced by Environmental Health – the same way the tattoo industry is regulated – to ensure that all premises and practitioners meet these standards over the coming years.”
Dr Martyn King, co-founder of Cosmedic Skin Clinic and medical director of ACE Group World, was party to the parliamentary discussion on the proposed new laws. He said: “Currently, dermal filler is classified as a medical device, which basically means that anybody can buy it. If this type of product was made prescriptiononly it would be a huge step to better regulation, ensuring only quality products are used and that practitioners have the medical background and training needed if complications should occur… It really would help to regulate the industry – at the moment, people are being put at huge risk.”
Meanwhile, Dr John Curran, recentlyappointed as head of The British College of Aesthetic Medicine’s (BCAM) Regulatory, Ethics and Professional Standards Committee, commented: “Having read the APPG report, it highlights the complete lack of regulation which allows anyone to carry out these procedures with minimal legal restrictions or qualifications, putting the public at great risk.
“We urge the government to prioritise making injectable fillers prescription-only and to review legislation around those who are permitted to administer such treatments.
“BCAM members, who are all qualified doctors and dentists, regularly see patients suffering complications from fillers administered by non-healthcare professionals who do not have the clinical expertise to carry out the treatments or access to the necessary prescription-only medicines when something goes wrong.”
NEWS IN BRIEF
Mount Snowdon climb for Alumier Foundation
Aesthetic Medicine editorial consultant Vicky Eldridge and AlumierMD education specialist Victoria Hiscock climbed Mount Snowdon to raise money for the Alumier Foundation. The pair, who are both directors of the foundation, tackled the Welsh mountain on 14 August to raise money to buy desks for new classrooms at Nhlengelo Primary School in White City, South Africa. The Alumier Foundation has so far enabled hundreds of students to further their education and bring light to their community.
Clinic launches covid travel portal
Multi-site aesthetic clinic Vivo Clinic has launched a comprehensive travel information portal to support its PCR testing service. The free portal allows travellers from the UK to find everything they need to know regarding covid-19 restrictions and requirements for over 330 international destinations, as well as general information on weather, currency and hotel availability. The clinic group has over 30 PCR test locations across the UK and Ireland. It also offers traffic light test packages that include all the tests needed to travel to amber and green countries.
Skinceuticals and Cutera form partnership
Clinical skincare brand Skinceuticals has joined forces with aesthetic technology manufacturer Cutera to develop a specialist treatment protocol to help optimise clinical outcomes for patients. In the protocol, Skinceuticals products will be used to prepare the skin for Cutera’s Laser Genesis procedure. Dr Julia Sevi will present the clinical benefits of this combined approach at Cutera’s University Clinical Forum on 27 September in Windsor. Register to attend: cutera.co.uk
Lynton to hold customer workshops
Lynton is helping accounts boost their business through customer workshops focusing on both marketing and clinical elements, with talks covering a range of topics. The first workshops are in Manchester on 20 September and London on 11 October. Demos of new launch Focus Dual will also take place. To book: lynton.co.uk