OVER 70S VOLUME RESTORATION
Gentle boost
Independent nurse practitioner and clinic director Yuliya Culley shares a case study of treating a patient in her early 70s with subtle collagen–stimulating filler
YULIYA CULLEY
Yuliya Culley (BSc, MSc, RGN, INP) is a registered general nurse and an independent nurse prescriber and founder of Novello Skin Clinic in Cheltenham. She has had experience in emergency, cardiology and dermatology nursing. and currently provides training for other medical practitioners through medical aesthetics supplier 4T Medical.
CONSULTATION
A 72-year-old female patient attended the clinic, initially due to flare ups of rosacea which have been an issue for her for years. The patient was very apprehensive to start any treatments as it was her first experience in a medical aesthetic clinic. However, she wished to improve her appearance to feel better overall. The patient is active, running her own successful business, and is perceived as a very sociable person. In general, she felt that not only her rosacea but also her facial volume needed addressing to achieve a fresher look.
After the patient experienced a positive improvement in her skin [see treatment plan below], and once we had established a trusted patient-clinician professional relationship, it was a suitable time to move to the next level of her treatment – to address her facial volume restoration. The patient asked about a suture lift, however, after taking into account all the facts including her age, collagen loss and moderate-severe soft tissue ptosis, she was advised to look into dual-action therapy such as Ellansé.1
This product has been recognised in the aesthetics medical field for its safety profile and collagen stimulation properties, via the process of neocollagenesis, dermal tissue thickening and, as a result, facial volume restoration through structural facial soft tissue support.4 This decision was made after initial consultation, a month before the first national lockdown in 2020. Once clinics were allowed to re-open, we organised a recap appointment to go through the treatment plan.
The preference was given to treatment with Ellansé M – which offers results for up to 24 months – out of three available products in the Ellansé portfolio.5 The patient was advised about Ellansé’s high safety profile however she was also told about possible reported side effects which include oedema for more than two weeks, bruising, molar oedema, temporary palpable lumps and discoloration.8 Other possible and less likely side effects associated with dermal filler injections such as infection, vascular compression, nodules and granulomas were discussed as a part of obtaining the patient’s informed consent with full transparency .8
While moderate volume loss in the temple area was observed, the patient decided against treatment in these areas for the first time. During midface assessment, the volume loss to the medial cheek area was identified as some asymmetry, representing more volume loss and skin crepiness to the left medial cheek due to malar volume reduction.2
There was also volume loss to the lateral aspect of the midface, inferiorly to the zygomatic arch. Both of these factors lead to development of heaviness around the nasolabial fat compartments with clinically more pronounced nasolabial folds and extended depression in the lid-cheek junction area.2
Lower-face assessment showed presence of jowls, pre-jowl sulcus, marionette folds and hyperactivity of mentalis, leading to chin shortening and presence of a horizontal chin line.
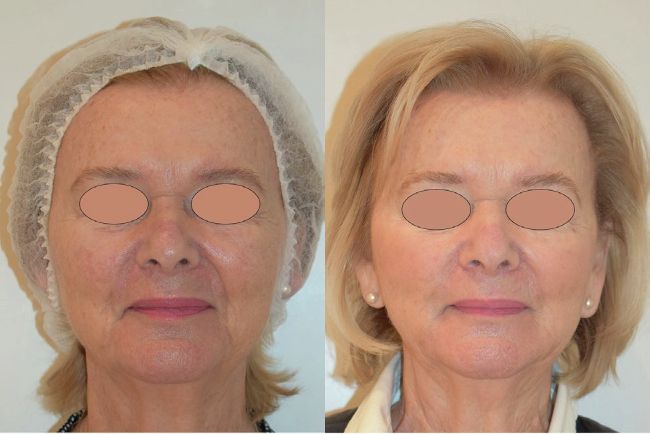
Left: before, Right: after
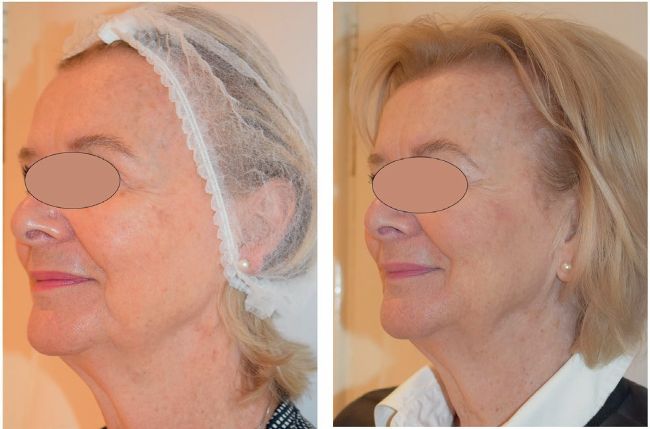
Left: before, Right: after
TREATMENT PLAN
Firstly, we created a bespoke plan which started with work to address the restoration of her skin barrier and telangiectasia reduction. The primary treatment plan included a course of eight Dermalux LED sessions, three sessions of Thermavein for facial telangiectasia reduction, and home skin care with ZO Hydrating Cleanser, ZO Renewal Moisturiser and an SPF50 product. In terms of the injectable portion of the overall treatment plan, the patient wished to look fresher with a reduction of overall skin sagginess on the face, with a conservative approach to facial rejuvenation and gradual results. The decision was made to divide the treatment plan into two sessions with a six-week gap to address volume loss and observe soft facial tissue reaction over the time thanks to the collagen-stimulating effect. This plan included the use of 4ml of Ellansé M during the first treatment and 3ml during the second, with further review at eight weeks after treatment completion.
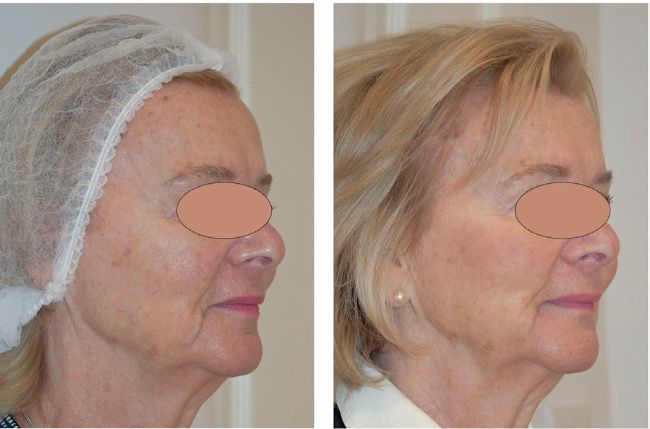
Left: before, Right: after
LMX 4 numbing cream was applied to areas of the mid and lower face for 30 minutes prior to the procedure to offer a more comfortable experien3,10 Skin was then prepared with Clinisept.9The areas of injections were marked with a water-soluble marker. The procedure was conducted with aseptic technique and use of a sterile pack 9 Ellansé M was mixed with 0.1ml of 2% lidocaine hydrochloride for each Ellansé syringe with 10 passes back and forth to obtain a homogenic solution of lidocaine and filer 6. A 25G 40mm cannula was inserted into the subcutaneous plane to introduce product using a retrograde fanning technique with each linear thread of 0.1ml/ cm2 for the purpose of safety and trauma reduction 5. This technique was used to address depression of the mid-medial cheek for structural soft tissue support to the area of the deficit.
0.55ml of product was used for the left and 0.45ml for the right medial cheek during the first treatment. For the second treatment, 0.45ml was used to the left and 0.3ml to the right medial cheek, respectively. 1ml was distributed to the lateral cheek bilaterally during both treatments, with a total amount of 2ml.
The lower-face treatment included marionettes, chin and nasolabial folds.7 The marionette area was treated with 0.5ml of product on each side during the first treatment and 0.4ml during the second, placed in the subcutaneous plane towards the chin midline bilaterally. The mental crease was injected using a linear threading technique in a retrograde manner, with 0.1ml injection placement on each side of the crease.⁵ The nasolabial folds were treated with 0.2ml of product on each side during the first treatment and 0.25ml during the second treatment with a subdermal fanning technique from the oral commissure ascending towards the cephalic extend of the nasolabial fold in a retrograde manner. This was done to avoid heaviness and an overfilled look. Direct treatment to the prejowl sulcus and jowls was not provided at this stage in order to achieve gradual results, and the plan was to improve these areas in the future after review at eight weeks upon completion of the initial treatment plan.
RESULTS
The patient attended a review eight weeks after the second treatment with an expression of gratitude as she felt more confident in her daily life, work and in socialising with her friends. Her friends observed that her skin looked fresher and smoother without knowing that a treatment had been performed. The patient became more confident to have further treatment to the pre-jowl sulcus and maintenance treatments in the future.
REFERENCES
1. Christe, M (2016) Ellanse Safety. A polycaprolactone-based collagen stimulator. Sinclaire Pharma: Paris. Available at: https://portal.sinclairpharma.com/portal/ ellanse/healthcare-physicians/clinical-studies [Accessed 2n April 2021].
2. Corey, C, Popelka, G, Barrera, J. and Most, S. (2012) An analysis of malar fat volume in two age groups: Implications for craniofacial surgery. Craniomaxillofacial Trauma and Reconstruction. 5 (4), pp. 231-234. Available at: https://www.ncbi.nlm.nih.gov/pmc/ articles/PMC3577599/ [Accessed 2nd April 2021].
3. Ferndale Pharmaceuticals Ltd (2019) LMX4 4% cream summary of product characteristics. Wetherby: Ferndale Pharmaceuticals Ltd. Available at: https://www. medicines.org.uk/emc/product/5938 [Accessed 2nd April 2021].
4. Kim,J (2019) Changes in dermal thickness in biopsy study of histologic findings after a single injection of polycaprolactone-based filler into the dermis. Aesthetic Surgery Journal. 39 (12), pp. NP484-NP494.
5. De Melo, F., Nicolau, P., Piovano, L., Lin, S., Baptista-Fernandes, T., King, M., Camporese, A., Hong, K., Khattar, M. and Christen, M. (2017) Recommendations for volume augmentation and rejuvenation of the face and hands with the new generation polycaprolactone-based collagen stimulator (Ellansé®). Clinical, Cosmetic and Investigational Dermatology. 10, pp. 431-440. Available at: https://www. ncbi.nlm.nih.gov/pmc/articles/PMC5685142/ [Accessed on 2nD April 2021].
6. De Melo, F. and Martijnissen-Hofste, J. (2012) Investigation of physical properties of a polycaprotactone dermal filler when mixed with lidocaine and lidocaine/ epinephrine. Dermatol Ther . 2(1):13. Available at: https://sinclair-college.com/pluginfile.php/5120/mod_resource/content/1/5ffd48afcc32f50a8c6ab62ef376039544af.pdf [Accessed 28th March 2021].
7. Hoschander, A., Salgado, J., Kassira, W. and Thaller, S. (2016) Operative procedures in plastic, aesthetics and reconstructive surgery.Chapter 16: Non-surgical facial rejuvenation with neuromodulators and dermal fillers. New York: Taylor and Francis Group.
8. Lin, S. and Christen, M (2020) Polycaprolactone-based dermal filler complications: A retrospective study of 1111 treatments. Journal of Cosmetic Dermatology. 19 (8), pp. 1907-1914. Available from: https://onlinelibrary.wiley.com/doi/full/10.1111/jocd.13518 [Accessed 1st April 2021].
9. Murthy, R., Eccleston, D., Mckeown, D., Parikh, A. and Shotter, S. (2020) Improving aseptic injection standards in aesthetic clinical practice. Dermatologic Therapies.34 (1), e14416. Available at: https://onlinelibrary.wiley.com/doi/full/10.1111/dth.14416 [Accessed 2nd April 2021].
10. Sobanko, J. Miller, C. and Alster,T (2012) Topical Anaesthetics for Dermatologic procedures:A review. Dermatologic Surgery. 38 (5), pp.709-721.